Dr. Guoliang Lu | 逯国亮

In all cells, transfer RNAs (tRNAs) convert the genetic information encoded in a messenger RNA (mRNA) into an amino acid sequence (protein). They relate converting three consecutive nucleotides of the mRNA (codon) to one amino acid at the ribosomal decoding and peptidyl transferase center. Since ribosomes were discovered in plant cells by Robinson and Brown (1953) and in animal cells by Palade (1955, who also coined the term “ribosome”), a wealth of biochemistry, cell biology, and structural biology studies expanded our knowledge on translational initiation, elongation, termination, and recycling.
We are currently interested in translational initiation in eukaryotes and its regulation by certain viral cis-acting elements (viral RNAs) and trans-acting factors (viral proteins). The control or deregulation of translational initiation in eukaryotes is involved in multiple disparate life phenomena, such as cancer, memory, and viral infection. More than ten initiation factors associate with the ribosome at various translational stages, allowing it to scan along mRNA. Viruses, especially RNA viruses, develop alternative translation regulation pathways, and some of them were eventually proved to be used by host cells.
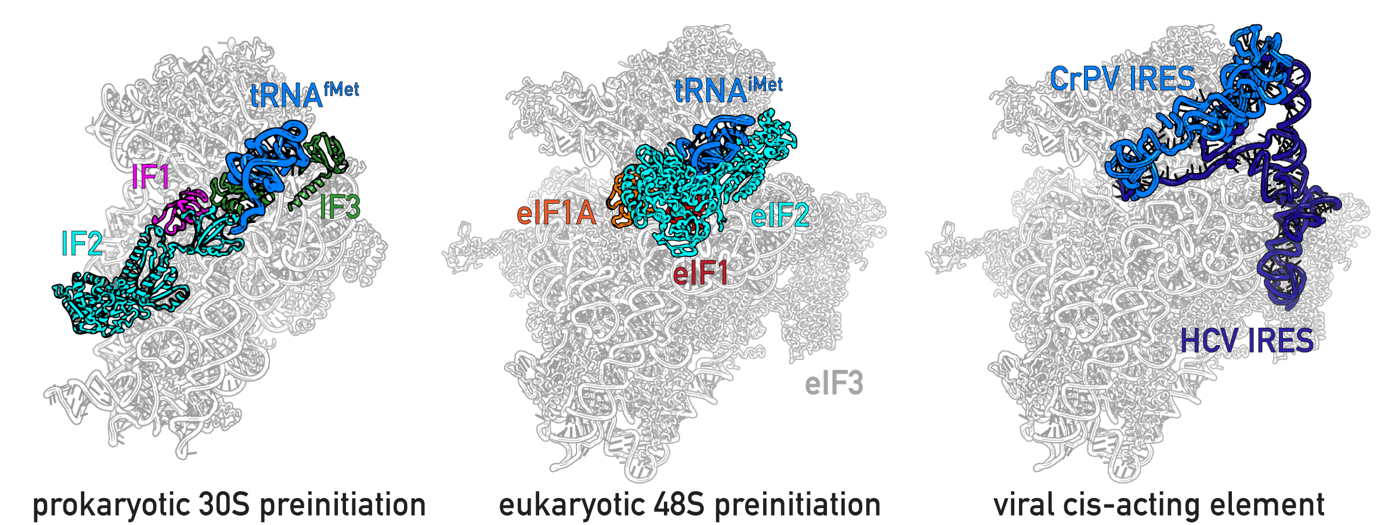
Translation initiation requires the formation of 30S/48S preinitiation complex (PICs), which involves the initiation factors and the initiator tRNA. Viral cis-acting element binds to host ribosome to initiate viral protein translation.
We are also interested in mRNA therapy. The worldwide spreaded SARS-CoV-2 developed into a global coronavirus disease pandemic coded COVID-19. We designed three mRNA vaccine candidates: receptor-binding domain (RBD), spike, and virus-like particles (VLP). The immunological evaluation showed that both the spike and VLP invoked robust immune responses in mice. A revised mRNA vaccine candidate expressing full-length spike antigen is now ready for clinical trials. Students who are interested in cell biology, mRNA therapy, and ribosome translation, please feel free to contact us. Hope to see you in the class and lab soon.
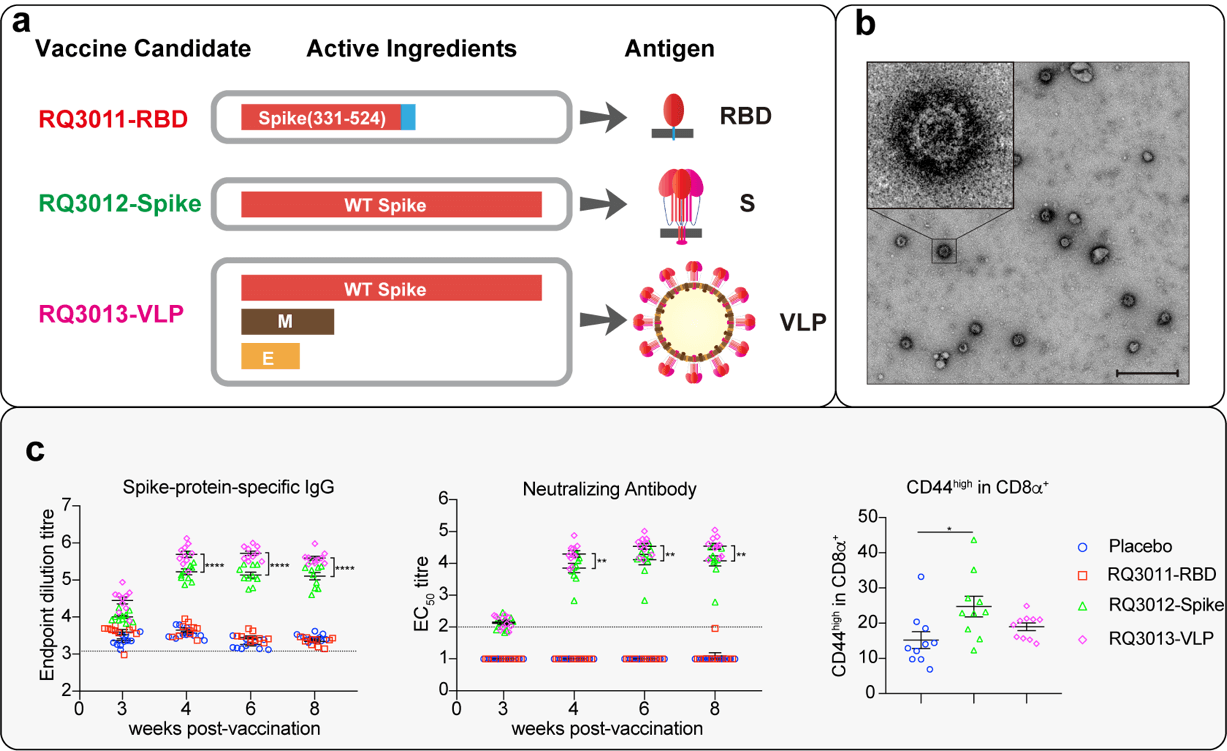
Humoral and cellular immune responses of mRNA LNP vaccine candidates. a) The three mRNA vaccine candidates for COVID-19. RQ3011-RBD contains an mRNA encoding the receptor binding domain of S with a C-terminal membrane-anchoring helix. RQ3012-Spike contains an mRNA encoding the full-length wild-type (WT) S. RQ3013-VLP contains three mRNAs encoding S, M, and E proteins that can assemble into VLPs. b) Electron microscopy images of VLPs produced by RQ3013-VLP. VLPs in the cell culture supernatant were purified and concentrated by ultracentrifugation and subjected to negative staining for electron microscopy. Scale bar, 500 nm. c) the antibody response (left panel) and neutralizing antibodies (center panel) were analyzed by S antigen and VSV-based pseudovirus assay, respectively. Frequency changes of CD8+ T cells 8 weeks after vaccination (right panel). Lines represent average titers of all animals in each vaccine group. The black dashed line indicates the limit of detection of the assay (reciprocal titer of 100).
